
Netmeds First Membership
Description
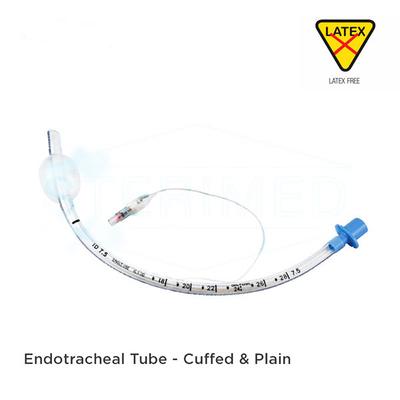
The Sterimed Endotracheal Tube Plain (SMD 701P) is a medical device designed for maintaining a patent airway and facilitating mechanical ventilation for patients. It is intended for short-term use in settings such as general anesthesia, intensive care, and emergency medicine. Constructed from a non-toxic, medical-grade PVC, this tube is designed to be kink-resistant and softens at body temperature to conform to the patient's respiratory tract anatomy. The plain design, without a cuff, is suitable for specific medical situations.
Thermo-sensitive Material: The tube is made from a thermo-sensitive material that softens at body temperature, allowing it to conform to the patient's airway for a better fit.
Atraumatic Design: Features a polished, smooth Murphy eye and a tapered distal end, which are designed to allow for smoother intubation and extubation with reduced tissue irritation.
Radio-Opaque Line: A radio-opaque line is incorporated into the tube's wall, enabling clear visualization of the tube's position on an X-ray to confirm correct placement.
Kink-Resistant Construction: The tube is designed to resist kinking, which helps to maintain an open and secure airway for the patient during use.
Standardized Connector: It is equipped with a universal 15mm connector at the proximal end, ensuring compatibility with standard ventilation equipment.
Q: What is the primary use of this endotracheal tube?
A: This tube is designed to be inserted through the mouth or nose into the trachea to maintain a clear airway for patients requiring mechanical ventilation in settings like general anesthesia or intensive care.
Q: What does 'Plain' signify for this endotracheal tube?
A: The term 'Plain' indicates that this endotracheal tube is uncuffed, meaning it does not have an inflatable balloon at its tip.
Q: Who is the 2.5 mm size intended for?
A: The 2.5 mm internal diameter size is typically used for neonatal patients, specifically newborns weighing less than 1000g or those under 28 weeks of gestation.
Q: Is this product sterile and for single use?
A: Yes, the endotracheal tube is supplied in a sterile, individually sealed peelable pouch and is intended for single-patient use to prevent cross-contamination.
Q: What material is the tube made from?
A: It is made from a non-toxic, medical-grade PVC material that is often siliconised, making it thermo-sensitive to soften at body temperature for patient comfort and to reduce trauma during insertion.
The Sterimed Endotracheal Tube Plain (SMD 701P) is a medical device designed for maintaining a patent airway and facilitating mechanical ventilation for patients. It is intended for short-term use ...
Key Benefits
Facilitates Airway Management: This endotracheal tube is used to establish and maintain a clear airway for patients who are unable to breathe on their own, often during surgery or in critical care.
Designed for Patient Comfort: The tube's material softens with body heat, and it has a smooth surface to ease the process of intubation, suctioning, and extubation.
Aids in Procedural Accuracy: The inclusion of a radio-opaque line allows healthcare professionals to verify the tube's placement radiologically, contributing to patient safety.
Facilitates Airway Management: This endotracheal tube is used to establish and maintain a clear airway for patients who are unable to breathe on their own, often during surgery or in critical care....
Direction for Use/Dosage
This product is intended for use by qualified healthcare professionals only. The tube is inserted through the patient's mouth or nose into the trachea to ensure the airway remains open for ventilation. The process is known as endotracheal intubation. It is recommended to lubricate the tube before insertion to minimize trauma. The position can be confirmed via radiological imaging, aided by the tube's radio-opaque line.
This product is intended for use by qualified healthcare professionals only. The tube is inserted through the patient's mouth or nose into the trachea to ensure the airway remains open for ventilat...
Safety Information/Precaution
For single-patient use only; do not reuse or re-sterilize as this may lead to cross-contamination.
This device should only be used by trained medical personnel familiar with endotracheal intubation procedures.
Inspect the packaging for any damage before use to ensure sterility is not compromised.
Always consult a healthcare professional for any medical advice or before using a medical device.
Possible side effects of intubation can include a sore throat or hoarseness after the tube is removed.
For single-patient use only; do not reuse or re-sterilize as this may lead to cross-contamination.
This device should only be used by trained medical personnel familiar with endotracheal intuba...
Other Information
The product is sterilized using Ethylene Oxide (E.T.O.) and is supplied pyrogen-free in a sterile, individually peelable pouch pack. Store in a cool, dry place away from direct sunlight. The material of construction is a non-toxic, non-irritant, medical-grade PVC, and it is Phthalate-free.
The product is sterilized using Ethylene Oxide (E.T.O.) and is supplied pyrogen-free in a sterile, individually peelable pouch pack. Store in a cool, dry place away from direct sunlight. The materi...





