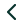

Netmeds First Membership
Description
The Polymed Polyflon Adva I.V. Cannula (24G) is a peripheral I.V. catheter designed for venous access to administer fluids, medications, or blood products. It features Adva needle technology, which provides a quick flashback to confirm successful venipuncture, enhancing the success rate on the first attempt. The catheter is made from biocompatible materials like FEP or polyurethane, which are designed for longer indwelling times and to reduce the risk of phlebitis.
Adva Needle Technology: Provides instant visual confirmation of blood flow along the catheter, which helps clinicians successfully access the vein on the first try.
Kink-Resistant Radiopaque Catheter: The catheter is made from flexible FEP/polyurethane, which resists kinking to ensure an uninterrupted flow of fluids and is visible on X-rays.
Flexible Wings: Perforated wings allow for secure fixation to the patient's skin, providing stability and reducing the risk of the cannula being dislodged.
Injection Port: Equipped with a non-return silicone valve, the port allows for the intermittent administration of medication without backflow.
Biocompatible Material: Manufactured from tested, biocompatible materials that are non-toxic and designed to allow for longer indwelling times with reduced patient discomfort.
Q: What is a 24G I.V. cannula used for?
A: A 24G I.V. cannula is a peripheral catheter used for intravenous access to administer fluids, medications, and blood products. Its smaller size makes it suitable for patients with smaller or more delicate veins, such as in pediatric or neonatal care.
Q: What are the functions of the wings on this cannula?
A: The flexible wings are designed to provide secure fixation to the skin, which helps in stabilizing the cannula and preventing it from dislodging during use.
Q: Is this cannula intended for single use?
A: Yes, this I.V. cannula is a sterile, single-use, disposable device. This is to minimize the risk of infections and cross-contamination between patients.
Q: What does the 'Adva' feature signify?
A: The 'Adva' needle technology provides a quick flashback feature, which allows for immediate visualization of blood flow upon successful venipuncture. This helps the clinician confirm correct placement and may enhance the success rate on the first attempt.
Q: Does this cannula contain a port for injections?
A: Yes, this type of cannula is typically equipped with an injection port that has a non-return valve, allowing for the administration of intermittent medication without needing a new puncture.
The Polymed Polyflon Adva I.V. Cannula (24G) is a peripheral I.V. catheter designed for venous access to administer fluids, medications, or blood products. It features Adva needle technology, which...
Key Benefits
The design facilitates a higher success rate in first-prick venipuncture, reducing patient discomfort and multiple insertion attempts.
Its biocompatible and flexible catheter material is designed to minimize trauma and reduce the chances of phlebitis, allowing for a longer indwelling time.
The integrated injection port allows for convenient and safe administration of medications without needing a separate needle.
Flexible wings provide secure and stable fixation, minimizing movement and potential displacement of the cannula.
The quick flashback feature offers immediate confirmation of correct vein entry, increasing procedural confidence for the healthcare professional.
The design facilitates a higher success rate in first-prick venipuncture, reducing patient discomfort and multiple insertion attempts.
Its biocompatible and flexible catheter material is design...
Direction for Use/Dosage
This product is intended for use by qualified healthcare professionals only. The site of insertion should be disinfected prior to cannulation. Follow established institutional protocols for I.V. cannula insertion, maintenance, and removal. After use, dispose of the cannula in accordance with medical waste disposal regulations.
This product is intended for use by qualified healthcare professionals only. The site of insertion should be disinfected prior to cannulation. Follow established institutional protocols for I.V. ca...
Safety Information/Precaution
For single use only. Do not reuse or re-sterilize, as this may lead to infection or malfunction.
Before use, inspect the individual sterile packaging for any signs of damage; do not use if the package is open or damaged.
This is a sterile product; handle with appropriate aseptic technique.
Use by a qualified medical professional is recommended.
The 24G size may not be suitable for the administration of highly viscous fluids or large blood transfusions.
Do not use in patients with known hypersensitivity to any of the materials used.
For single use only. Do not reuse or re-sterilize, as this may lead to infection or malfunction.
Before use, inspect the individual sterile packaging for any signs of damage; do not use if the ...
Other Information
Store in a cool, dry place, away from direct sunlight. The 24G cannula is identifiable by its yellow color code. The catheter has a length of 19 mm and an outer diameter of 0.7 mm, with a flow rate of approximately 23 ml/min. The product is free from PVC, DEHP, and Latex.
Store in a cool, dry place, away from direct sunlight. The 24G cannula is identifiable by its yellow color code. The catheter has a length of 19 mm and an outer diameter of 0.7 mm, with a flow rate...