Netmeds First Membership
Introduction About PHENERGAN
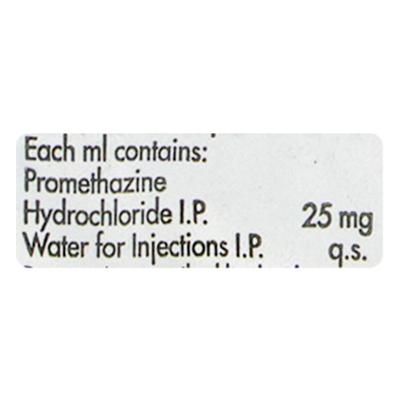
Phenergan Injection 2ml contains promethazine, an antihistamine-based injectable formulation that is used in clinical practice under strict medical supervision. It is generally administered in hospital or healthcare settings where injectable forms are required for structured symptom support. This medicine is not meant for self-use and is only given by trained healthcare professionals in controlled environments.
This injection is commonly considered in situations involving nausea, vomiting, allergy-related symptoms, motion-related discomfort, or sedation-related clinical support needs. It is typically used when oral medicines are not suitable or when faster clinical response is required under medical evaluation. The decision to use this formulation is based on patient condition, symptom severity, and overall clinical assessment by a healthcare provider.
Phenergan Injection 2ml works as part of the antihistamine class, where promethazine interacts with histamine-related pathways and also influences central nervous system responses. This combined pharmacological activity helps provide structured symptom support in controlled medical care environments. It is usually administered either intramuscularly or intravenously by healthcare professionals depending on clinical requirements and hospital protocols.
This injection is suitable for patients who require supervised injectable antihistamine support in clinical settings. It should be used with caution in individuals with certain medical conditions, and a full medical history review is recommended before administration. During pregnancy and breastfeeding, it is only considered if clearly advised by a healthcare professional after evaluating the risk–benefit balance. Self-administration is not recommended under any circumstances.
Common responses after administration may include drowsiness, dizziness, fatigue, or mild injection site reactions in some individuals. These effects are generally temporary and vary based on individual sensitivity and clinical condition. Continuous monitoring in healthcare settings helps ensure appropriate response and safe administration. Regular medical supervision and adherence to clinical guidance are important for appropriate use of this injectable formulation.
Uses Of PHENERGAN
It is used to manage:
• Allergic reactions (itching, hives, angioedema)
• Severe allergic reactions (as an adjunct in anaphylaxis)
• Allergic reactions to blood or plasma transfusion
• Motion sickness
• Nausea and vomiting (including drug-induced and post-operative)
• reduction of nausea and vomiting associated with anaesthesia and surgery
• Sedation and anxiety relief
• Pre-operative, post-operative, and obstetric (labour) sedation
• Post-operative pain (as an adjunct to analgesics)
• As an adjunct to anaesthesia in special surgical procedures
Benefits of PHENERGAN
Support for Nausea and Vomiting–Related Discomfort
Phenergan Injection 2ml may be used to support relief from nausea and vomiting–related discomfort. It helps manage multiple uneasiness sensations that can occur in clinical care environments. It is generally used under healthcare supervision for short-term symptom support.
Benefits of Antihistamine-Based Injectable Formulations
Antihistamine injectable formulations offer support by acting on histamine-related responses in the body. They provide broader symptom support compared to some single-action approaches. Such formulations are commonly used under professional medical guidance in clinical environments.
Use of Phenergan Injection 2ml for Nausea-Related Discomfort
Phenergan Injection 2ml may help support individuals experiencing nausea-related discomfort in clinical care settings. It assists in managing uneasiness that may affect physical comfort and stability. It is generally used in monitored healthcare situations.
Support in Motion Sickness–Related Discomfort
This injection may be used to support relief from motion sickness–related discomfort. It helps manage travel-associated uneasiness and imbalance sensations. It is administered under medical supervision when clinically considered appropriate.
Support in Controlled Clinical Symptom Environments
Phenergan Injection 2ml is used in controlled healthcare environments where symptom-based support is required. It helps manage multiple discomfort sensations under monitored conditions. It is administered as part of clinical decision-making by healthcare professionals.
How PHENERGAN Works
How Promethazine Interacts with Histamine H1 Receptors
Promethazine works by interacting with histamine H1 receptors in the body, helping regulate histamine-related responses in a controlled physiological way. This interaction is part of its antihistamine classification used in clinical pharmacology explanations.
Antihistamine Receptor Blocking Action Explained
Promethazine is known for its ability to block histamine H1 receptor activity at a cellular level. This receptor-level interaction is a key concept in understanding how antihistamine compounds function within the body.
Central Nervous System Activity of Promethazine
Promethazine also shows activity in the central nervous system, where it interacts with neural pathways involved in alertness and sensory response. This contributes to its overall pharmacological profile in medical science.
Sedating Antihistamine Mechanism in Pharmacology
In pharmacological terms, promethazine is categorized as a sedating antihistamine due to its action on both histamine receptors and central nervous system pathways. This dual interaction is widely described in medical reference texts.
Receptor-Level Activity of Phenergan Injection
At the receptor level, Phenergan Injection 2ml demonstrates binding interactions that influence histamine signaling. This receptor-based activity forms the foundation of its pharmacological classification.
How to use PHENERGAN
Recommended Dosage Guidelines for Phenergan Injection 2ml
- Dosage is determined in clinical settings based on individual assessment
- Administered only under healthcare supervision
- Adjustments are made according to patient-specific clinical evaluation
- Used as part of structured medical care protocols in hospitals
Intramuscular Administration Method Explained
- Given as an injection into muscle tissue by healthcare professionals
- Commonly used in controlled clinical environments
- Requires sterile handling and proper injection technique
- Performed only in healthcare settings
Intravenous Administration Process in Clinical Settings
- Administered directly into a vein under supervision
- Conducted in monitored healthcare environments
- Requires controlled infusion or slow administration as per clinical standards
- Handled only by trained medical personnel
Dosage Considerations in Clinical Practice
- Based on individual health evaluation
- Depends on clinical condition and response factors
- Determined only by qualified healthcare professionals
- Adjusted according to standard medical protocols
Step-by-Step Injection Administration Overview
- Patient evaluation is performed by healthcare professional
- Appropriate route (intramuscular or intravenous) is selected
- Injection is prepared under sterile conditions
- Administration is carried out using clinical technique
- Post-administration observation may be conducted in healthcare settings
Side Effects Of PHENERGAN
Common side effects include:
• Drowsiness (sedation)
• Dizziness
• Blurred or double vision
• Confusion or disorientation
• Dry mouth
• Fatigue or weakness
• Nausea or vomiting
• Constipation or diarrhoea
• Loss of appetite
• Injection site reactions (pain, swelling)
• Low blood pressure (hypotension) or faintness
• Increased or irregular heart rate (tachycardia/bradycardia
When to Consult Your Doctor:
• Severe allergic reaction (wheezing, difficulty breathing, swelling of face/lips/tongue)
Warning & Precautions
Pregnancy
Consult your doctorPhenergan Injection should be used during pregnancy only if clearly needed. High doses in late pregnancy may cause movement disorders (extrapyramidal symptoms) in the newborn. Limited human data are available, so the potential benefits must outweigh risks. Always consult your doctor before use.
Breastfeeding
Consult your doctorPhenergan Injection is not recommended during breastfeeding unless clearly prescribed by your doctor. It may pass into breast milk in small amounts. Although serious harmful effects are uncommon, caution is advised. Infants should be monitored for side effects such as excessive sleepiness or sedation. Always consult your doctor before using this medicine.
Driving and Using Machines
Use with CautionPhenergan Injection may cause marked drowsiness and impair mental and physical abilities. Avoid driving or operating machinery until you know how the medicine affects you.
Alcohol
Consult your doctorAlcohol should be avoided while taking Phenergan Injection, as it can significantly increase sedation and CNS depression. This may lead to severe drowsiness or impaired coordination.
Kidney
Use with CautionNo specific data available, but caution is advised in patients with impaired organ function. Consult your doctor before use.
Liver
Use with CautionPhenergan Injection should be used with caution in patients with liver impairment or jaundice. Concomitant use with other hepatotoxic medicines may increase the risk of liver damage. Regular monitoring may be required. Always consult your doctor before taking this medicine.
Allergy
ContraindicatedPhenergan Injection contains sodium metabisulfite, which may cause allergic reactions, including severe reactions (anaphylaxis) and asthma attacks in susceptible individuals. Seek immediate medical help if symptoms occur.
Lungs
ContraindicatedPhenergan Injection is not recommended in patients with compromised respiratory function (e.g., COPD, sleep apnea).
Heart Disease
Consult your doctorPhenergan Injection should be used with caution in patients with cardiovascular disorders. It is not recommended without medical supervision as it may cause hypotension and QT interval prolongation, increasing the risk of serious heart rhythm disturbances. Always consult your doctor before taking this medicine.
Use In Pediatrics
Consult your doctorPhenergan Injection is contraindicated in children under 2 years of age due to the risk of fatal respiratory depression. Use with caution in children aged 2 years and above. It may cause serious adverse effects such as hallucinations, seizures, or, in rare cases, sudden death at high doses. Avoid use in children with suspected Reye’s syndrome or liver disease. Always consult your doctor before taking this medicine.
Use In Geriatrics
Consult your doctorElderly patients may be more sensitive to sedative effects. Lower doses are recommended with careful monitoring.
Other Warnings for PHENERGAN
Before Taking This Medicine, Inform Your Doctor If You Have:
- Epilepsy or seizure disorders
- Bone marrow disorders
- Glaucoma
- Prostate enlargement
- Gastrointestinal obstruction
- History of severe allergic reactions
Drug - Drug interaction
CNS Depressants (e.g., alcohol, barbiturates, narcotics, sedatives, tranquilizers, general anesthetics)
Interaction: Promethazine enhances the sedative effects of CNS depressants.
Effects: Increased sedation, respiratory depression, prolonged drowsiness.
Management: Avoid combination or reduce doses (barbiturates ↓ by at least 50%, narcotics ↓ by 25–50%). Monitor patient closely.
Epinephrine (Adrenaline)
Interaction: Promethazine may reverse the vasopressor effect of epinephrine.
Effects: Ineffective management of hypotension; possible worsening low blood pressure.
Management: Do NOT use epinephrine in cases of hypotension due to promethazine overdose.
Anticholinergic Agents (e.g., atropine, antihistamines)
Interaction: Additive anticholinergic effects when used together.
Effects: Dry mouth, constipation, urinary retention, risk of paralytic ileus.
Management: Use cautiously; report gastrointestinal symptoms immediately.
Monoamine Oxidase (MAO) Inhibitors (e.g., phenelzine, selegiline)
Interaction: Increased risk of CNS depression and extrapyramidal symptoms.
Effects: Hypotension, severe sedation, movement disorders.
Management: Avoid or use with caution under medical supervision.
Anticonvulsants (e.g., phenytoin, valproate)
Interaction: Promethazine may lower seizure threshold.
Effects: Increased risk of seizures.
Management: Dose adjustment of anticonvulsants may be required.
Antihypertensive Agents (e.g., propranolol, beta-blockers)
Interaction: May inhibit metabolism, increasing drug levels.
Effects: Additive hypotension, arrhythmias, possible retinal and movement disorders.
Management: Monitor blood pressure and adjust doses as needed.
Guanethidine
Interaction: Inhibits neuronal uptake of guanethidine.
Effects: Reduced antihypertensive effect.
Management: Monitor blood pressure; alternative therapy may be needed.
Bromocriptine
Interaction: Promethazine increases prolactin levels, opposing bromocriptine action.
Effects: Reduced effectiveness of bromocriptine.
Management: Dose adjustment may be necessary.
Levodopa
Interaction: Dopamine receptor blockade reduces levodopa effectiveness.
Effects: Worsening Parkinson’s symptoms.
Management: Avoid concurrent use if possible.
Metrizamide
Interaction: Lowers seizure threshold when used together.
Effects: Increased risk of seizures.
Management: Stop promethazine 48 hours before and restart 24 hours after procedure.
Phenothiazine Derivatives
Interaction: Additive extrapyramidal effects.
Effects: Increased movement disorders.
Management: Avoid combination or monitor closely.
Quinidine
Interaction: Additive cardiac effects.
Effects: Risk of arrhythmias.
Management: Use cautiously with cardiac monitoring.
Sympathomimetic Agents (e.g., adrenaline, amphetamines, ephedrine)
Interaction: Promethazine blocks alpha-adrenergic effects.
Effects: Hypotension, tachycardia, reduced stimulant effects.
Management: Avoid combination or monitor cardiovascular status.
Tricyclic Antidepressants (e.g., amitriptyline)
Interaction: Increased anticholinergic and sedative effects.
Effects: Hypotension, CNS depression, extrapyramidal symptoms.
Management: Use cautiously; monitor patient closely.
Sedative Herbal Products (e.g., valerian, kava)
Interaction: May enhance sedative effects.
Effects: Increased drowsiness and CNS depression.
Management: Avoid or use with caution; consult doctor before use.
Synopsis
|
Drug |
Promethazine |
|
Pharmacological Category |
Antihistamine (H1 receptor antagonist) with sedative and antiemetic properties |
|
Therapeutic Indication |
Used to manage allergic conditions (such as itching, rashes, hay fever), reduce and control nausea and vomiting, manage motion sickness, and provide sedation (e.g., before or after procedures) |
|
Dosage Forms |
Injection (2 ml ampoule) |
More Information
1) Why are injectable antihistamines being discussed more in modern hospital safety practices?
Growing Focus on Standardized Hospital Protocols
Hospitals are placing more emphasis on structured and standardized injectable medication practices to ensure consistent clinical handling.
Improved Documentation Systems
Digital health records and pharmacy systems are making it easier to track injectable medicine use and administration details.
Emphasis on Clinical Training and Awareness
Healthcare teams are focusing on regular training programs to strengthen understanding of injectable formulations and their handling.
Clear Separation of Dosage Forms
There is increased attention on distinguishing between oral and injectable medication formats for better clinical clarity.
Patient Safety Communication Improvements
Hospitals are improving how they communicate medication-related information in simple, understandable formats.
Focus on Controlled Clinical Environments
Injectable antihistamines are being reviewed within the context of controlled administration settings and supervised care practices.
2) How does this safety discussion improve understanding of injectable antihistamine use in hospitals?
Better Clinical Decision Clarity
Healthcare professionals rely on updated safety discussions to support structured and informed clinical choices.
Enhanced Awareness of Administration Processes
These conversations help reinforce how injectable medicines are handled within hospital workflows.
Strengthened Professional Guidelines
Updated discussions contribute to refined clinical reference guidelines used in healthcare settings.
Improved Patient Education Approach
Patients benefit from clearer explanations about how injectable medications are managed in clinical care environments.
Greater Focus on Controlled Handling
There is stronger emphasis on safe storage, preparation, and administration practices in hospitals.
Support for Evidence-Based Practice
These discussions encourage reliance on clinical references and established medical protocols.
Increased Transparency in Healthcare Communication
Hospitals are adopting clearer communication practices to help individuals better understand injectable medication processes.
FAQs About PHENERGAN
Q: What is Phenergan Injection 2 ml used for?
A: Phenergan Injection 2 ml contains Promethazine, which is commonly used under medical supervision to support relief from allergy-related symptoms such as itching, runny nose, and skin reactions. It is also used to manage nausea, vomiting, and motion-related discomfort in clinical settings.
Q: How does Phenergan Injection work in the body?
A: It works by blocking histamine activity in the body and affecting certain signals in the brain. This helps reduce allergic responses and supports control of nausea and vomiting sensations.
Q: How is Phenergan Injection given?
A: It is administered by a healthcare professional through intramuscular or intravenous injection. The dosage and method of administration depend on the patient’s condition and medical assessment.
Q: Can Phenergan Injection make you sleepy?
A: Yes, drowsiness or sedation is a common effect due to its action on the central nervous system. Patients are usually advised to rest after administration.
Q: Who should use caution before receiving Phenergan Injection?
A: Individuals with breathing problems, liver conditions, glaucoma, or those taking sedative medicines should inform their doctor before use. Medical history is important before administration.
Q: Is Phenergan Injection used during pregnancy or breastfeeding?
A: It should only be used during pregnancy or breastfeeding if clearly advised by a healthcare professional after assessing potential risks and benefits.
Q: What are the common side effects of Phenergan Injection?
A: Common effects may include drowsiness, dizziness, dry mouth, or mild injection site discomfort. These effects are usually temporary.
Q: Can I drive after receiving Phenergan Injection?
A: It is generally advised to avoid driving or operating machinery after receiving this injection due to possible drowsiness and reduced alertness.
Q: How quickly does Phenergan Injection work?
A: The onset of action depends on the route of administration and individual response, but effects are generally observed within a short time after administration.
Q: Can Phenergan Injection be used for motion sickness?
A: Yes, it may be used in clinical settings to help manage motion-related nausea and vomiting under medical supervision.
Q: What should I do if I experience unusual reactions after injection?
A: Any unusual or persistent symptoms should be reported immediately to a healthcare professional for proper evaluation and guidance.
References
1.KD Tripathi. Antiemetic, Prokinetic andDigestant Drugs. Drugs Essentials of medical pharmacology. 8th edition. 2013. Page –711
2. Hikma Pharmaceuticals USA Inc. PHENERGAN- promethazine hydrochloride injection. [Revised date6/2025]. [Accessed date 31/03/2026] ![]()
3. PDR by connective RX.Phenergan. [Accessed date 31/03/2026] 
4. Hospira Australia Pty Ltd.Promethazine Hydrochloride Injection BP.[Revised date February 2026] [Accessed date 31/03/2026]