
Netmeds First Membership
Description
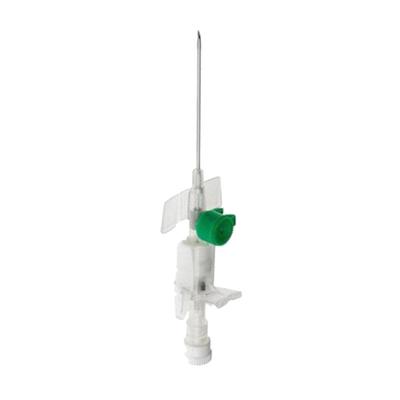
The Becton Dickinson (BD) Venflon Pro Safety IV Cannula is a peripheral intravenous catheter designed to provide reliable and safe venous access. It is specifically engineered to reduce the risk of accidental needlestick injuries and blood spatter, enhancing safety for healthcare professionals. This cannula is suitable for a variety of clinical applications, including rehydration, the administration of medication, parenteral nutrition, and blood transfusions. It is constructed with BD Vialon™ biomaterial, which is designed to soften once inside the vein, potentially minimizing complications.
Safety Mechanism: Incorporates a passive safety feature that automatically covers the needle tip as it is withdrawn, which helps to guard against needlestick injuries. BD Vialon™ Catheter Material: The catheter is produced from a distinct biomaterial that softens within the vein, potentially permitting longer dwell times and lowering the risk of mechanical phlebitis. Injection Port: Features a ported design that includes an integrated snap cap to help prevent it from opening accidentally. Ergonomic Design: The grip is structured to accommodate a variety of insertion techniques, offering flexibility to clinicians. Back-cut Needle Design: The needle is configured for proficient skin and vein penetration.
Q: What is the main purpose of the BD Venflon Pro Safety IV Cannula?
A: This is a peripheral IV catheter designed for safe and reliable intravenous access to administer fluids or medications, featuring a system to protect against needlestick injuries.
Q: What does the 'Safety' feature in this cannula do?
A: It incorporates a passive safety mechanism where a shield automatically covers the needle tip upon withdrawal, which helps to minimize the risk of accidental needlestick injuries for healthcare professionals.
Q: What does the '18G' in the product name signify?
A: The '18G' refers to the gauge of the cannula, which indicates its size. An 18G cannula has a green color code for easy identification of its size.
Q: Is this cannula intended for single use?
A: Yes, this is a sterile medical device intended for single use only and should be disposed of in a sharps container after use. This practice is vital for preventing infections and ensuring patient safety.
Q: Who should use this product?
A: This medical device is intended for use by trained healthcare professionals for intravenous cannulation procedures. It is advisable to consult a healthcare provider for any medical procedures.
The Becton Dickinson (BD) Venflon Pro Safety IV Cannula is a peripheral intravenous catheter designed to provide reliable and safe venous access. It is specifically engineered to reduce the risk of...
Key Benefits
The BD Venflon Pro Safety IV Cannula offers several advantages for both clinicians and patients. Its primary benefit is the automated needle-shielding mechanism, which helps protect healthcare workers from needlestick injuries and potential exposure to bloodborne pathogens. The catheter material is formulated to soften inside the vein, which can support longer indwelling periods and may decrease the incidence of phlebitis. The design also aims to minimize blood spatter during and after activation, contributing to a cleaner and safer clinical environment. For the clinician, the ergonomic grip and specialized needle design facilitate various insertion methods and aid in smooth vein penetration. Additionally, the integrated injection port with a snap cap allows for the convenient administration of medications and fluids.
The BD Venflon Pro Safety IV Cannula offers several advantages for both clinicians and patients. Its primary benefit is the automated needle-shielding mechanism, which helps protect healthcare work...
Direction for Use/Dosage
Always adhere to the Aseptic Non-Touch Technique (ANTT) throughout the procedure. Disinfect the intended insertion site according to your facility's established protocol. Carefully remove the cannula from its sterile packaging and lower the wings. Grip the device using your preferred method, such as placing fingers on the wings or employing a straight technique on the needle grip. Insert the cannula into the vein at a low angle, watching for blood return in the flashback chamber. Lower the device until it is nearly parallel to the skin and advance the entire system a small distance (approximately 2-3 mm) to confirm the catheter tip is securely inside the vein. Keep the needle stationary while advancing the catheter completely into the vein. Stabilize the catheter, apply pressure to the vein just beyond the catheter tip, and release the tourniquet. Withdraw the needle; the safety mechanism is designed to engage automatically to cover the needle tip. Properly dispose of the sharp in a designated container.
Always adhere to the Aseptic Non-Touch Technique (ANTT) throughout the procedure. Disinfect the intended insertion site according to your facility's established protocol. Carefully remove the cannu...
Safety Information/Precaution
This product is intended for single use only; do not reuse. It is designed for use by trained healthcare professionals. Always follow local guidelines and institutional policies for IV cannulation and the disposal of sharps. It may be advisable to replace the cannula routinely every 72 hours if it remains in use, or as directed by facility protocol. Do not use the product if the sterile packaging is damaged or has been compromised. The safety feature activates upon needle withdrawal to help prevent accidental needlestick injuries. It is recommended to consult a healthcare professional for proper use.
This product is intended for single use only; do not reuse. It is designed for use by trained healthcare professionals. Always follow local guidelines and institutional policies for IV cannulation ...
Other Information
The product is supplied sterile and is latex-free. The 18G cannula is identified by its green color, features a needle length of 45mm, and has a flow rate of approximately 103 ml/min. This cannula is compatible with both BD Luer-Slip and Luer-Lock connectors. The catheter is appropriate for use with power injectors set to a maximum pressure of 325 psi (2240 kPa). Store in a cool, dry location, away from direct sunlight.
The product is supplied sterile and is latex-free. The 18G cannula is identified by its green color, features a needle length of 45mm, and has a flow rate of approximately 103 ml/min. This cannula ...





