
Netmeds First Membership
Description
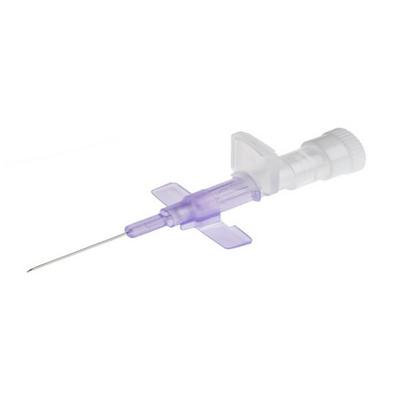
The Becton Dickinson BD Neoflon Pro IV Cannula is a device intended for accessing small and fragile vessels with precision. It is constructed with BD Vialon™ biomaterial and incorporates BD Instaflash™ needle technology to provide visual confirmation of vessel entry. This design is suitable for various insertion techniques and aims to enhance patient comfort and procedural efficiency. The cannula also features elements to improve cannulation control and clinician safety.
Precision Vessel Access: Designed to access smaller, fragile vessels with minimal trauma, utilizing BD Instaflash™ needle technology.
Ergonomic Design: Suited for all insertion techniques, featuring an anti-rotation tab for enhanced control and tactile feedback during insertion.
Visual Confirmation: The Instaflash™ needle technology provides immediate visual confirmation of successful vessel entry, which may reduce multiple insertion attempts.
BD Vialon™ Biomaterial: The cannula is made from a proprietary material that softens in the vein, allowing for longer dwell times and reducing the risk of mechanical phlebitis.
Safety Features: Incorporates a passive needle shield that automatically covers the needle after cannulation to aid in preventing needlestick injuries.
Q: What is the primary use of the BD Neoflon Pro IV Cannula?
A: This cannula is designed for intravenous access, particularly for smaller and more fragile veins, such as those in neonatal, pediatric, and geriatric patients. It should only be used by a qualified healthcare professional.
Q: What do the specifications 26G, 0.6mm, and 19mm signify?
A: 26G refers to the gauge of the needle, which indicates its diameter. 0.6mm is the outer diameter of the cannula, and 19mm is its length. This small size is intended for use in delicate vessels.
Q: What is the cannula made from?
A: It is made from a proprietary material called BD Vialon™ biomaterial, which is designed to soften inside the vein, potentially reducing the risk of mechanical phlebitis.
Q: Does this cannula have any features for confirming vein entry?
A: Yes, it incorporates BD Instaflash™ needle technology, which provides a visual confirmation of successful vessel entry, helping to reduce multiple insertion attempts.
Q: Is this product intended for single use?
A: Yes, the BD Neoflon Pro IV Cannula is a sterile, single-use medical device to ensure patient safety and prevent cross-contamination.
The Becton Dickinson BD Neoflon Pro IV Cannula is a device intended for accessing small and fragile vessels with precision. It is constructed with BD Vialon™ biomaterial and incorporates BD Instafl...
Key Benefits
Provides access to small and fragile veins.
Designed to reduce the risk of vessel trauma and improve patient comfort during intravenous therapy.
Visual flashback technology helps confirm successful vessel entry.
The use of BD Vialon™ biomaterial may allow for longer catheter dwell times.
Incorporates features to protect against needlestick injuries.
Provides access to small and fragile veins.
Designed to reduce the risk of vessel trauma and improve patient comfort during intravenous therapy.
Visual flashback technology helps confirm su...
Direction for Use/Dosage
For use by a qualified healthcare professional. Prepare the insertion site as per facility policy. Stabilize the vessel and perform venipuncture at a low angle. Upon observing initial blood return in the catheter, lower the angle to be almost parallel to the skin and advance the catheter and needle unit slightly further. Hold the needle stationary and advance the catheter off the needle completely into the vein. Release the tourniquet, stabilize the catheter hub, and withdraw the needle, disposing of it in a sharps container. Connect accessory devices as needed and secure the catheter. Always consult the product label and follow institutional protocols.
For use by a qualified healthcare professional. Prepare the insertion site as per facility policy. Stabilize the vessel and perform venipuncture at a low angle. Upon observing initial blood return ...
Safety Information/Precaution
This product is intended for use by trained healthcare professionals only. Handle with care. Read the product label carefully before use. Never reinsert the needle into the catheter as this could shear the catheter. After use, dispose of the cannula and needle in a puncture-resistant, leak-proof sharps container according to local policy. Store in a cool, dry place away from direct sunlight. Keep out of reach of children.
This product is intended for use by trained healthcare professionals only. Handle with care. Read the product label carefully before use. Never reinsert the needle into the catheter as this could s...
Other Information
Country of Origin: India. The product is sterile and for single use only. The material is latex-free. The cannula is made of BD Vialon™, a polyurethane biomaterial. The 26G cannula has a purple color code.
Country of Origin: India. The product is sterile and for single use only. The material is latex-free. The cannula is made of BD Vialon™, a polyurethane biomaterial. The 26G cannula has a purple col...